|
|
Vol.
29 No. 6
November-December 2007
Quantities, Units, and Symbols in Physical Chemistry: 3rd edition—also known as the IUPAC Green Book
prepared for publication by E.R. Cohen, T. Cvitas, J.G Frey, B. Holmstrom, K. Kuchitsu, R. Marquardt, I. Mills, F. Pavese, M. Quack, J. Stohner, H. Strauss, M. Takami, and A.J. Thor
RSC Publishing, 2007
ISBN 0 85404 433 7; ISBN-13 978 0 85404 433 7
The first IUPAC Manual of Symbols and Terminology for Physicochemical Quantities and Units (the Green Book) of which this is the direct successor, was published in 1969, with the object of “securing clarity and precision, and wider agreement in the use of symbols, by chemists in different countries, among physicists, chemists and engineers, and by editors of scientific journals.” In 1988, it underwent major extension and revision and was given the simplified title Quantities, Units and Symbols in Physical Chemistry.
This third edition of the Green Book, Quantities, Units, and Symbols in Physical Chemistry, is based upon the most up-to-date sources for fundamental constants, data, and nomenclature in the fields of chemistry and physics. As with previous versions, this edition has been written to enable clear understanding in an interdisciplinary environment and to convey information in a global multidisciplinary arena in which chemistry plays a central role. Examples are provided to explain how to avoid ambiguity in conveying
information.
The third edition has a significantly extended index that provides a dictionary of terms and symbols and useful conversion tables. Information in the Green Book is synthesized from IUPAC, IUPAP, and ISO. The second edition has been available online as a PDF file, and the third edition will be available as a PDF one year after publication.
This is the definitive guide for scientists and organizations working across a multitude of disciplines requiring internationally approved nomenclature.
www.iupac.org/publications/books/author/cohen.html
Not convinced that the Green Book is an invaluable reference to have? Try the quiz below . . .
- What is the symbol for amount of substance (also called chemical amount)?
- What is the definition of the SI base unit for amount of substance, the mole?
- What is the affinity of a chemical reaction?
- How is it defined, and what is its SI unit?
- Define the extent of reaction
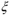 (SI unit: mole) for a chemical reaction? (SI unit: mole) for a chemical reaction?
- What is the SI unit, and the commonly used unit, of molar electric conductivity?
- What do the acronyms RADAR, REMPI, RHEED, and STEM stand for?
- Name 12 different quantities for which the symbol μ is commonly used?
- Define the number-average, mass-average, and Z-average molar masses of a polymer.
- How is the reduced spin-spin coupling KAB in NMR spectroscopy related to the more familiar (indirect) spin-spin constant JAB? Why is KAB sometimes a more useful quantity?
- The magnetic susceptibility of a sample is described as “-15 cgs ppm.” Convert this into SI units.
- Give the present definition of the metre.
- The integrated absorption intensity of the
1479 cm-1 band ion of the infrared spectrum of benzene is recorded to be 2.34 km mol-1. Convert this (i) into an integrated cross-section in pm2, and (ii) into an intensity in atm-1 cm-2 at 20°C.
- The étendue of an optical instrument is a measure of its throughput, or light gathering power. Define fluence; give its symbol, and SI units.
- Define
the coulomb integral Jij and
the exchange integral Kij between
two molecular orbitals øi
and øj in Hartree-Fock
SCF-MO theory.
- There are two accepted definitions of the electronegativity χ of an atom; give them both. In what units is χ usually recorded?
- What are the meanings of the units ppb and ppt? When should they not be used?
- Give the SI prefixes for 10-15, 10-18, 10-21, and 10-24.
- What is the definition of the year?
- Will the year 3200 be a leap year?
Compiled by Ian Mills (Vol 16, No. 1. 1994, CI) following the publication of the second edition of the Green Book in 1993.
Page
last modified 26 November 2007.
Copyright © 2003-2007 International Union of Pure and Applied Chemistry.
Questions regarding the website, please contact [email protected]
|