ORGANIC CHEMISTRY
DIVISION
COMMISSION ON NOMENCLATURE OF ORGANIC CHEMISTRY
Phane Nomenclature.
Part I: Phane Parent Names
(Recommendations 1998)
W.H. Powell
World Wide
Web version prepared by G. P. Moss
Synopsis: Phane nomenclature is a new
method for building names for organic structures by assembling names
that describe component parts of a complex structure. It is based on
the idea that a relatively simple skeleton for a parent hydride can
be modified by an operation called "amplification", a process that replaces
one or more special atoms (superatoms) of a simplified skeleton by multiatomic
structures. In this set of recommendations, multiatomic structures are
fully saturated rings or ring systems or unsaturated rings or ring systems
with the maximum number of noncumulative double bonds (mancude). In
the amplification operation each superatom is replaced by a ring or
ring system identified in the name by an "amplification prefix" attached
to a stem called a "simplified skeletal name". The latter ends with
the term "phane" and is formed according to the principles for deriving
names of saturated hydrocarbons. Accordingly, all of the atoms implied
by the skeletal name, except for the superatoms are, by convention,
traditional saturated carbon atoms. An amplification prefix is derived
from the name of the corresponding cyclic parent hydride by the addition
of the terminal letter "a" with elision of a terminal vowel of the parent
hydride name, if present. Phane prefixes thus resemble the traditional
skeletal replacement ("a") prefixes, such as "oxa", "aza", etc., that
indicate replacement of a single skeletal unit of structure, usually
a carbon unit, by a different unit of structure.
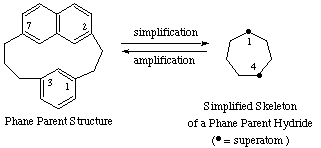
Simplified skeletal name: cycloheptaphane
Phane parent hydride name: 1(2,7)-naphthalena-4(1,3)-benzenacycloheptaphane
The locants in front of the parentheses in the phane parent hydride
name identify the positions of the superatoms in the simplified skeleton
that are replaced by the ring structure specified by the amplification
prefix immediately following. By the same token, they also identify
the positions of the rings and ring systems in the phane parent hydride.
These locants are determined by the inherent numbering of the simplified
skeleton and the seniority of the rings and ring systems in the phane
parent hydride. The locants within the parentheses specify the atoms
of the ring structure expressed by the amplification prefixes that are
linked to the adjacent normal atoms of the simplified parent skeleton.
In addition to the basic principles of phane nomenclature, Part I
contains the fundamental methodology for numbering substituents and
the application of skeletal replacement ("a") nomenclature for naming
heterophane parent hydrides.
World Wide
Web version prepared by G. P. Moss
http://www.chem.qmw.ac.uk/iupac/phane/

